US FDA Services Group LLC may also assist with compliance in international markets.

FDA registration is required for all facilities that manufacture, process, pack, or store food, beverages, or dietary...
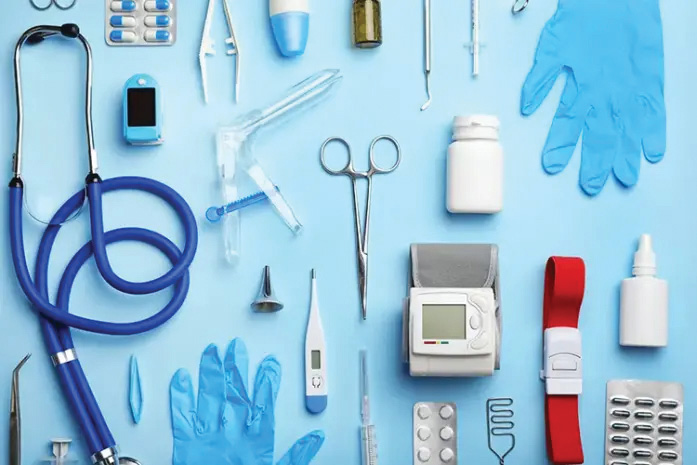
Companies who are involved in manufacturing, testing, packing, labeling, sterilization, or import of medical devices...

US FDA Services Group LLC is a premier regulatory consultancy based in Wyoming, USA, serving as a strategic bridge for global manufacturers to the United States market. We specialize in providing comprehensive compliance solutions for the food, drug, medical device, and cosmetic industries, with a dedicated focus on exporters in Pakistan, India, Bangladesh, and the broader South Asian region.
US FDA Services Group LLC provides end-to-end support for international manufacturers entering the U.S. market. With a professional team backed by years of industry experience, we simplify the path to compliance for food, medical device, drug, and cosmetic exports.
US FDA Services Group LLC may also assist with compliance in international markets.




Get a quote by emailing, or filling out the form.
For any registration inquiries, don’t hesitate to message us. We’ll be happy to assist you!
The U.S. FDA Registration Renewal period CLOSED on December 31, 2024
If you did not renew by the deadline, you must re-register with FDA.
Re-register or verify that your registration was renewed for 2025-2026: